

INTRODUCTION
Many household cleaning problems are blamed on weak products or poor technique, but in reality, the issue often comes due to pH. Whether a cleaner is acidic, neutral, or alkaline determines what types of dirt it can remove, how it interacts with surfaces, and whether it poses any kind of safety risks during use.In UK and US households, where water hardness, surface materials, and cleaning habits vary widely, understanding pH is essential for effective and safe cleaning. A product with the wrong pH may clean poorly, damage surfaces, or create unnecessary health risks.This article explains what pH means in the context of cleaning chemistry, how pH affects cleaning performance, and why matching pH to the task is more important than choosing stronger chemicals.
What Is pH?
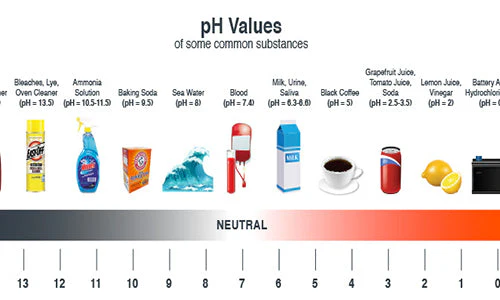
In generic term, pH is a measure of how acidic or alkaline a substance is, expressed on a scale from 0 to 14.
pH below 7on the pH scale indicates acidity
pH exactly of 7 on the pH scale is neutral
pH above 7 on the pH scale indicates alkalinity
In cleaning products, pH directly influences chemical reactivity, soil removal mechanisms, and material compatibility.
Why pH Matters in Cleaning Chemistry
So, as cleaning is a chemical process, here different types of soil respond to different chemical conditions, and pH controls how cleaning agents interact with those dirt & soils.
pH and Greasy or Oily Soil
Grease and oily soils respond best to alkaline conditions. Alkalinity helps break down fats through emulsification and saponification and allowing surfactants to remove them more effectively.
This is closely linked to how surfactants actually work in household cleaning products, where pH influences micelle formation and soil suspension.
pH and Mineral-Based Soil
Mineral deposits such as lime scale are alkaline in nature and thus require acidic cleaners todissolve them. This is why acidic formulations are used in bathrooms and kitchens to address scale buildup.
The chemistry behind this is explored further when discussing lime scale formation and removal in household cleaning.
Acidic Cleaners: Where They Work Best
Acidic cleaning products typically have a pH between 1 and 6. They are effective against:
Lime scale and mineral deposits
Soap scum
Rust staining
However, acidic cleaners can damage certain surfaces, including natural stone and some metals,
if used excessively. So, understanding surface compatibility is also critical, especially in hard
water regions where scale removal is frequent.
Alkaline Cleaners: Strengths and Limitations
Alkaline cleaners usually have a pH between 8 and 14. They are effective at:
Removing grease and oils
Breaking down protein-based soils
Supporting surfactant performance
But on the other side, high alkalinity, however, increases the risk of the following:
Fabric damage
Surface etching
Skin irritation
This explains why overdosing detergent damages fabrics and washing machines and why stronger the cleaner is not always better for your clothings.

Neutral Cleaners and When They Are Used
Neutral cleaners, with a pH close or equal to 7, are designed for:
Delicate surfaces
Frequent cleaning
Areas where safety and material preservation are priorities
They are often used on sealed floors, finished wood, and sensitive materials, even though their cleaning power may be lower than acidic or alkaline products. But here material sensitivity took more into account than aggressive and rigid cleaning.
pH, Water Hardness, and Detergent Performance
Water chemistry significantly affects how pH behaves during cleaning. In hard water areas, calcium and magnesium ions can buffer or alter effective pH levels during washing. This interaction explains how water hardness affects detergent performance and why builders are necessary to stabilise cleaning conditions.
pH and Enzyme Stability in Modern Detergents
Enzymes used in detergents function optimally within narrow pH ranges as these are highly pH sensitive. Outside these ranges, enzyme activity drops sharply, reducing stain removal efficiency. This relationship becomes particularly important in cold wash systems, discussed further in why
detergents perform differently in cold wash cycles.
Safety Risks Associated with Extreme pH
Products with very low or very high pH can:
Cause skin and eye irritation
Damage respiratory tissues
React dangerously when mixed
This is why understanding pH is essential for household safety and why mixing household cleaners can be dangerous to your cloths and surfaces as well as to your skin.


Why pH Alone Does Not Define Cleaning Performance
While pH is important but it does not operate in isolation. Effective cleaning essentially requires the following ingredients into the cleaning products:
Appropriate surfactants
Builders to control water hardness
Enzymes for biological soils
Correct formulation balance
A high or low pH without a comprehensive chemical formulation support rarely delivers consistent results.
Conclusion
pH is a defining factor in how cleaning products perform and how safely they can be used. Acidic and alkaline cleaners each have specific roles, and using the wrong pH for a given task leads to poor results and potential damage. In UK and US households, where water hardness and surface diversity are common challenges, understanding pH allows for informed product selection and safer cleaning practices. Matching
pH to the task, rather than choosing stronger formulations, remains the foundation of effective cleaning chemistry.
