Surfactants are the functional backbone of almost every household cleaning product, including laundry detergents, dishwashing liquids, and surface cleaners. Despite their widespread use, theway surfactants actually remove dirt andgreaseis often misunderstood or oversimplified.
Effective cleaning is not driven by fragrance, foam, or colour. It is driven by chemistry.
In UK and US households, differences in water hardness, soil composition, and surface materialssignificantly affect how surfactants behave during cleaning. Understanding these interactions explains why some products perform consistently while others fail under identical conditions.This article explains how surfactants function at a chemical level, how they interact with soils,and why formulation design matters more than marketing claims.
What Are Surfactants?
Surfactants, short for surface-active agents, are chemical compounds designed to reduce surface tension between two unlike substances. In cleaning systems, they allow water to interact with oils, greases, and particulate soils that would otherwise repel it.
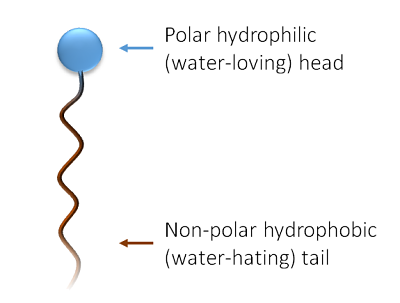
At a molecular level, surfactants consist of:
A hydrophilic head that interacts with water and
A hydrophobic tail that interacts with oils and grease
This dual structure allows surfactants to act as a bridge between water and oily soils, making
removal possible during washing and rinsing.
(IMAGEs for SURFACTANT STRUCT
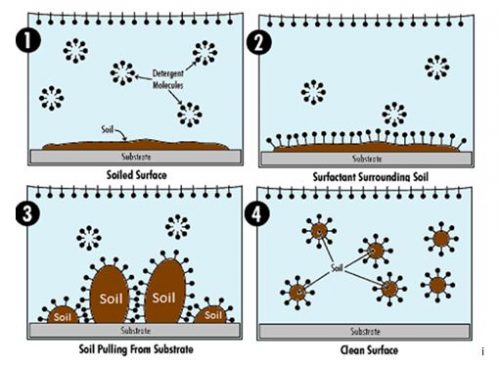
How Surfactants Remove Dirt and Grease
Surface Tension Reduction
Pure water has high surface tension, limiting its ability to spread across surfaces and penetrate soil. Surfactants reduce this surface tension, allowing water to wet fabrics and hard surfaces more effectively. This effect is particularly important in regions where mineral-rich water
already reduces cleaning efficiency, a topic explored further when discussing that how water hardness affects detergent performance.
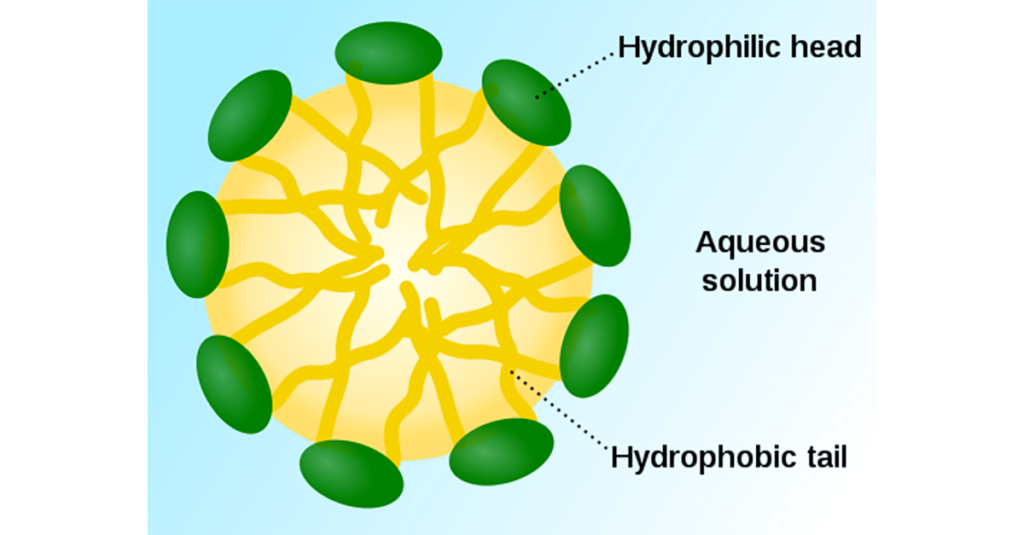
Micelle Formation and Soil Encapsulation
When surfactant concentration reaches the critical micelle concentration, surfactant molecules
form structures called micelles.
Inside a micelle:
Hydrophobic tails trap oily soil at the center and;
Hydrophilic heads remain oriented toward water.
This process allows grease and oil to be lifted from surfaces and suspended in water rather than
redeposited during rinsing.
Types of Surfactants Used in Household Cleaning Products
Anionic Surfactants
Anionic surfactants are widely used in laundry detergents and dishwashing liquids due to their strong grease-removal capability. However, they are sensitive to calcium and magnesium ions found in hard water.
This sensitivity explains why choosing the correct detergent type is critical in hard water areas.
Nonionic Surfactants
Nonionic surfactants are less affected by water hardness and remain effective at lower temperatures. They are commonly used in modern liquid detergents and cold-wash formulations.
Amphoteric and Cationic Surfactants
Amphoteric surfactants are used in mild cleaning systems, while cationic surfactants are primarily used for fabric conditioning and antimicrobial functions rather than primary soil removal.
Why Foam Is Not a Measure of Cleaning Power
Foam is a visible side effect of certain surfactants, not an indicator of cleaning performance. High foam does not mean better soil removal and can interfere with rinsing efficiency, especially in modern washing machines. This misunderstanding is addressed in detail when explaining that
why more foam does not mean better cleaning.
Impact of Water Chemistry on Surfactant Performance
Hard Water Interference
In many UK and US regions, household water contains significant levels of calcium and magnesium ions. These ions interact with surfactants, reducing micelle formation and overall cleaning efficiency. This interaction contributes to residue formation and inconsistent results,
which is why understanding the water hardness and detergent chemistry is essential.
Temperature Effects
Temperature influences surfactant solubility, micelle formation, and soil removal kinetics. Lower temperatures reduce reaction rates unless formulations are specifically designed for cold-water performance.
Why Surfactants Alone Are Not Enough
Surfactants operate as part of a formulation system. Effective cleaning products combine
surfactants with builders, enzymes, solvents, and stabilisers to ensure performance under varying
conditions.
For example:
Enzymes target protein, fat, and starch-based soils
Builders support surfactant performance in hard water
Solvents assist with stubborn greasy residues
Understanding this balance explains why single-ingredient solutions rarely perform consistently.
Conclusion
Surfactants are the backbone of household cleaning chemistry. Their ability to reduce surface tension, form micelles, and interact with soils determines whether a product cleans effectively or fails.
However, surfactant performance is inseparable from water chemistry, formulation design, and application conditions. In UK and US households, where water hardness and usage patterns vary widely, understanding these interactions explains why informed product selection leads to better
cleaning outcomes.
